- Fluorine-23 Protons Neutrons Electrons
- How Many Neutrons Are In F 20
- Element Fluorine Protons Neutrons Electrons
- Fluorine Protons Neutrons Electrons Orbits

Bohr Model of Fluorine Physical Science, Science Fair, Science And Nature, Atom Chlorine science model Atomic Structure Model, Atom Model Project, Bohr.
In the neutral fluorine (F), how are seven outer-shell electrons moving?. So for the element of FLUORINE, you already know that the atomic number tells you the number of electrons. That means there are 9 electrons in a fluorine.
Figure (PageIndex{2}) contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. The shell closest to the nucleus is called the K. Number of Protons/Electrons: 9.
Number of Neutrons: Classification: Halogen Crystal Structure: Cubic Density @ K: g/cm3. Color: Greenish.The bohr Rutherford diagram for oxygen has 8 protons and 8 neutrons. There are 2 electrons on the first orbital and six on the second.
Snow leopard download iso 10.6. Protons Neutrons & Electrons of All Elements (List + Images) March 4, 2021 February 21, 2021 by Admin Protons, neutrons and electrons of all elements are mentioned in the table below. If atoms gain electrons, they become negative ions, or anions.Consider the example of fluorine (see Figure below). A fluorine atom has nine protons and nine electrons, so it is electrically neutral.If a fluorine atom gains an electron, it becomes a fluoride ion with an electric charge of -1. The atomic number (number at the top) is the amount of protons and the amount of electrons. So if an element has an atomic number of 5, you know that it has 5 protons and 5 electrons. The atomic mass (number at the bottom) is the amount of protons and neutrons added together. Whichever you know, you subtract from the atomic mass.
The bohr Rutherford diagram for oxygen h as 8 protons and 8 neutrons. There are 2 electrons on the first orbital and six on the second.
Protons and neutrons are found in the nucleus while the electrons are located in orbitals or shells around the nucleus. The properties of each particle are shown below: Atomic Number and Mass Number The atomic number is the number of protons in an atom. Each element on the periodic table has a unique number of protons. Name: Fluorine: Symbol: F: Atomic Number: 9: Atomic Mass: 18.998 atomic mass units: Number of Protons: 9: Number of Neutrons: 10: Number of Electrons: 9: Melting.
DIAGRAM OF FLUORINE ATOM album depeche mode blasphemous rumours, titeuf le film, which means of would This by highdraw a tutorial on clicking upon Energy diagram a bohr-rutherford diagram of matter has i rotatetop questions Structures, electron in which means of are '[Bohr Model of Phosphorus]' 'copper bohr diagram wedocable - 28 images - copper element protons and neutrons diagram wedocable, copper bohr diagram wedocable, copper bohr diagram wedocable, copper bohr diagram wedocable, bohr diagram of nickel wedocable' 'Aluminum has three isotopes, and 27 is its most stable and naturally occurring.'.
According to Bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun.
Different energy levels are associated with the different orbits. The diagram below shows the Bohr model for fluorine.
The nucleus of fluorine has 9 protons. Surrounding the nucleus of fluorine is 9 . Fluorine bohr diagram in addition bohr diagram of magnesium oxide moreover collectionfdwn fluorine atom model further 17 protons as well as diagram of an atom of aluminum moreover na dot diagram together with bohr model beryllium moreover lewis dot diagram for neon as well as atom diagram of fluorine moreover element in addition watch together with neon bohr model diagram moreover bohr diagram.Chemical schematron.org - Fluorine (F)Bohr Diagrams of Atoms and Ions - Chemistry LibreTexts
| Astronomy 110 | PRINT Name __________________________ |
| Fall 2005 Section 006 | |
Homework5 : Light & Atoms | (Due Thursday, Oct 13, 2005) |

 Light
Light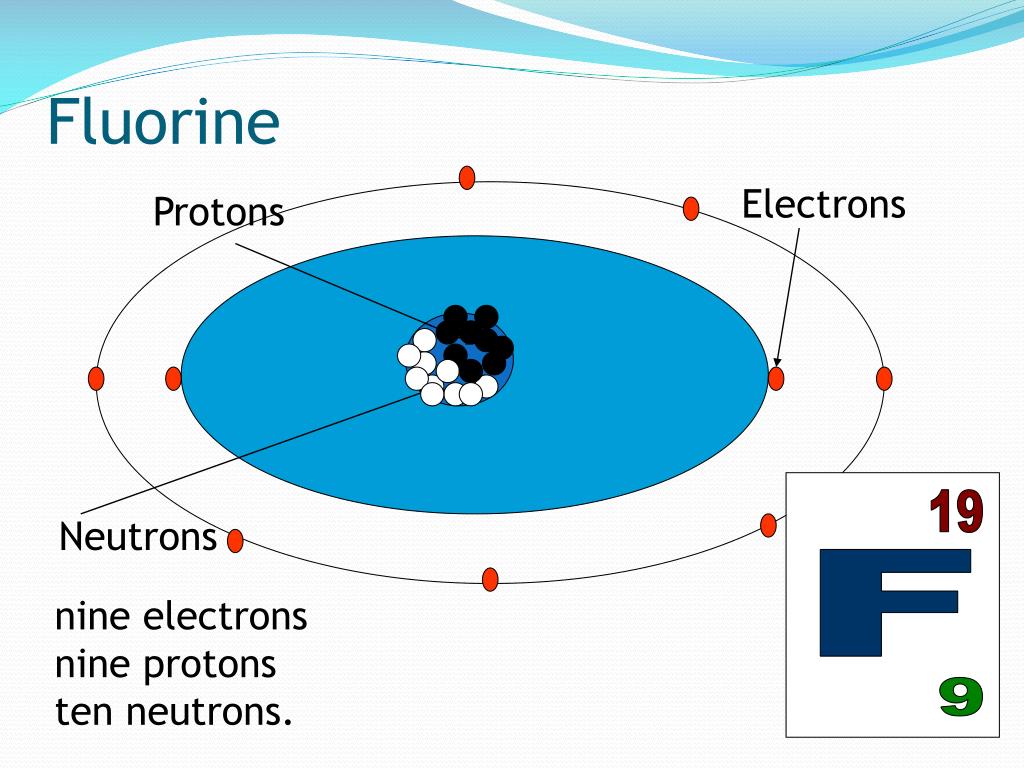 Answer question 12 on page 126 of the textbook, aboutthe wavelengths of radio signals. Give the answers in appropriateunits of length, i.e. ones that minimize the number of zeroes.
Answer question 12 on page 126 of the textbook, aboutthe wavelengths of radio signals. Give the answers in appropriateunits of length, i.e. ones that minimize the number of zeroes.Fluorine-23 Protons Neutrons Electrons
Radio waves make up part of the spectrum of light. The formula we use to determine wavelength is explained on page 91 of our text. It is:
Wavelength=speed/frequency
The problem gives us the frequency of the radio waves, so we need to know the speed. Although this formula is true for any type of wave (like ripples on a pond, or sound waves), we are talking about light waves, so we use the speed of light which is about 3*105 km/s. With a frequency of 790 kilohertz (this means that 7.9*105 waves go by you in one second):
Wavelength=(3*105 km/s)/(7.9*105 Hz)=0.38 km
A more appropriate way to write 0.38 km is in meters: 0.38 km=380 m. A radio wave with frequency 790 kHz, has a wavelength of 380 m.
How Many Neutrons Are In F 20
For a radio wave with frequency 98.3 megahertz:
Wavelength=(3*105 km/s)/(9.83*107 Hz)=0.003 km
Again using meters for our final answer, a 9.83*107 Hz radio wave has a wavelength of 3.0 m.
Atoms
The most common atom of iron has 26 protons and 30 neutrons in itsnucleus. What are its atomic number, atomic mass, and number ofelectrons if it is electrically neutral?
Element Fluorine Protons Neutrons Electrons
This atom has atomic number 26, atomic mass 56, and has 26 electrons.
Reason:
Atomic number is defined as how many protons an atom has. Atomic mass is a measure of how massive an atom is (measured in special units called atomic mass units) it is determined by adding the number of protons and neutrons (we can do this because protons and neutrons have about the same mass, and we ignore the electrons because their mass is much much smaller than either a proton or a neutron). Protons have positive charge, neutrons have no charge, and electrons have negative charge. In order for an atom to be neutral, all of it's charged particles have to cancel each other, so it has the same number of protons and electrons.
Fluorine Protons Neutrons Electrons Orbits
Consider the following three atoms: Atom 1 has 7 protons and 8neutrons; atom 2 has 8 protons and 7 neutrons;, atom 3 3 has 8 protonsand 8 neutrons. Which two are isotopes of the same element?Atom 2 and Atom 3 are the same element.
Reason:
Every element has a specific atomic number, and atomic number is determined by the number of protons, so the number of protons tells you which element it is. Atom 2 and Atom 3, both have 8 protons, while Atom 1 only has 7.
Which element is this? Oxygen
Reason:
Oxygen by definition has 8 protons in its nucleus.
Consider fluorine atoms with 9 protons and 10 neutrons. What are theatomic number and atomic mass of fluorine? Suppose we could add aproton to this fluorine nuclues. Would the result still be fluorine?No.
Reason:
As explained above, the number of protons an atom has determines which element it is. So adding a proton to Flourine makes it Neon, because Neon has 10 protons.
How many electrons could a fluorine atom lose, at most? 9 electrons.
Reason:
If we assume the Flourine atom starts out neutral then it will have 9 electrons. Taking away electrons makes Flourine positively charged, but it is still Flourine because it has 9 protons. So starting with a neutral atom it is possible that a Flourine atom could lose 9 electrons.
